Evidence-Led Strategy Across the Product Lifecycle
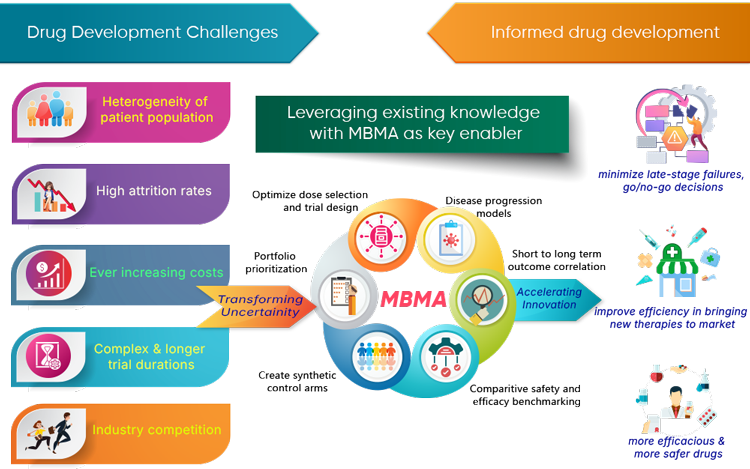
Modern drug development demands more than clinical progression — it requires early, data-driven decisions that reduce uncertainty and strengthen strategic positioning. Evidence is the key enabler for rigorous quantitative thinking early and consistently, using which organisations can navigate uncertainty, make informed trade-offs, and create long-term value.
EviGenuity brings its rich experience and processes to build on-time, reliable and comprehensive evidence datasets that support decision making throughout the product life cycle.
“Accelerating information access to enable efficient drug development and health technology evaluation”
One stop source for your Clinical & Real world evidence
Data is the key aspect of the rational approaches, and we offer a suite of evidence services across several therapeutic areas that include oncology, autoimmune, metabolic & cardiovascular, neuroscience, pain & inflammation and others all through the clinical development to market access.